eOrganic authors:
Olivia M. Smith, School of Biological Sciences, Washington State University
William E. Snyder Ph.D., Department of Entomology, Washington State University
This is one in a series of three articles about insectivorous birds on organic farms. The other articles are:
Introduction
A growing body of experimental evidence suggests that birds play important roles as natural enemies in agricultural ecosystems. For example, a study conducted in Europe demonstrated the important services provided by Great Tits (Parus major) in apple orchards. Researchers experimentally added nest boxes to some plots and saw an increase in fruit yield from 4.7 to 7.8 kg per tree. Increased yield was attributed to predation of caterpillars by Great Tits (Mols et al., 2002). A review paper by Bael et al. (2008) found that across 48 studies examined, birds reduced arthropods and plant damage. Here we focus on identification, diet, and management of Western Bluebirds and their role in farming. In addition to the insects this species consumes, we make suggestions for how to manage bluebirds to maximize their benefits while minimizing their risks.
- Identification
- Diet
- Habitat During the Growing Season
- Management
- More Resources
- References and Citations
Identification

Figure 1. Western Bluebird male holding an insect. Photo credit: Nigel Winnu, Western Bluebird, CC Attribution 2.0

Figure 2. Western Bluebird female. Photo credit: Martin Jambon, Female Western Bluebird, CC Attribution 2.0 Generic
Western Bluebirds (Sialia mexicana) are in the thrush family along with the familiar American Robin (Turdus migratorius) and the possibly less familiar Swainson's Thrush (Catharus ustulatus). Western Bluebird males are strikingly blue on the head, neck, wings, and tail. A rust-orange belt crosses the breast, with a slightly duller blue on the belly than the wings (Fig. 1). The brilliant blue on males is replaced by a duller blue-gray on females and juveniles (Fig. 2). The similar Mountain Bluebird (Sialia currucoides) lacks the rusty breast and flank plumage of the Western Bluebird, and the Mountain Bluebird is more sky blue (Fig. 3). The Eastern Bluebird (Sialia sialis) is also similar in appearance to the Western Bluebird, but its rust color extends up the throat, and it lacks blue on the breast (Fig. 4). The Eastern Bluebird occurs further east than the Western Bluebird; however, the Western Bluebird has been expanding eastward over the last several decades and displacing Eastern Bluebirds due to greater aggression and high dispersal ability (see Fig. 5 for a range map; Duckworth and Badyaev, 2007). Another similar species is the Lazuli Bunting (Passerina amoena), but the Lazuli Bunting has prominent wing bars, a smaller body size, and a thicker bill (Fig. 6).

Figure 3. Mountain Bluebird. Photo credit: Nigel Winnu, Nigel Winnu, Mountain Bluebird, CC Attribution 2.0

Figure 4. Eastern Bluebird. Photo credit: Jeff Bryant, Eastern Bluebird, CC Attribution 2.0
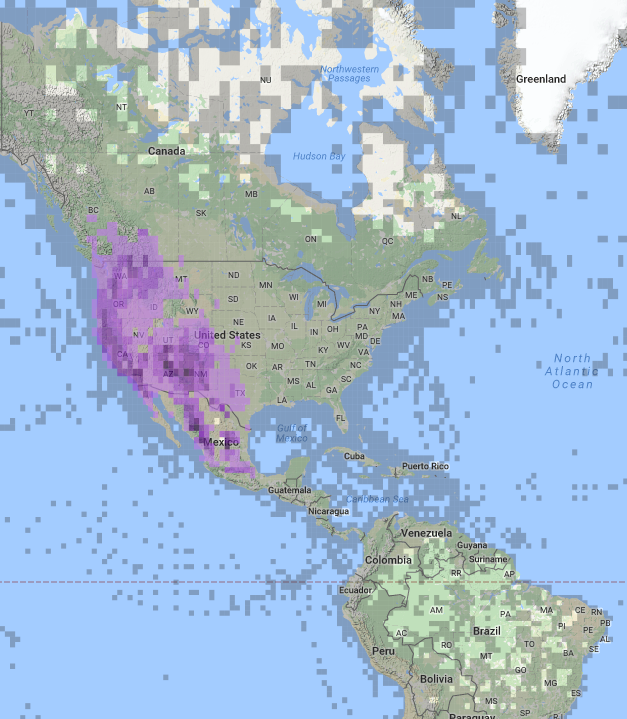
Fig 5. Range map for the Western Bluebird from eBird. [Image provided by eBird (www.ebird.org) and created 10 January 2017]

Fig 6. Lazuli Bunting. Photo credit: Julio Mulero, Lazuli Bunting, CC Attribution-NonCommercial-NoDerivs 2.0
Diet
Western Bluebirds are primarily insectivorous ground gleaners (De Graaf et al., 1985) and often forage off of perches. Grasshoppers and beetles may be the most important portion of the nestling bluebird diet (Beal, 1915; Herlugson, 1982). A study conducted in South-central Washington examined the diet of bluebird adults and chicks in the breeding season. The nestling diet was composed of 37.5% Coleoptera (beetles), 29.2% Hymenoptera (wasps, bees), 17.5% Hemiptera (true bugs including aphids and scales), 9.4% Orthoptera (grasshoppers), 2.5% Lepidoptera (caterpillars), and 1.0% Arachnida (spiders). Actual biomass of each taxa in the nestling diet differed slightly than the number of individuals consumed: 58.27% Orthoptera, 22.85% Homoptera, 10.42% Coleoptera, 4.45% Arachnida, 3.80% Lepidoptera, and 0.21% Hymenoptera. The adult diet differed slightly from the nestling diet. The primary constituents of the adult diet were beetles (68.2%) and caterpillars (12.2%). After nestlings hatched, the diet shifted to Coleoptera (37.5%), Hymenoptera (29.2%), and Hemiptera (15.6%) (Herlugson, 1982). Beal (1915) additionally found flies and snails in gut contents. A study using a novel technique called molecular scatology tested the DNA in Western Bluebird feces and found that Aedes mosquitos comprised the highest portion of the diet (present in 49.5% of samples). Ectoparasitic bird blowfly (Protocalliphora sp.) DNA was in 7% of adult and 11% of nestling samples. Herbivorous insects from the Hemiptera and Lepidoptera orders comprised 56% of the prey items in bluebird diets. Predatory insects and parasitoid insects were less than 3% of the diet, suggesting that Western Bluebirds offer substantial ecosystem services and little risk of consuming beneficial predatory arthropods (Jedlicka et al., 2017).
Western Bluebirds have been shown to provide ecosystem services in California organic vineyards. The study experimentally added nest boxes to some vineyard sites and compared avian species richness, Western Bluebird abundance, and beet armyworm (Spodoptera exigua) predation in sites with added nest boxes and without nest boxes. The study found that with addition of nest boxes, the average species richness of avian insectivores increased by over 50%. Further, density of insectivorous birds quadrupled, and Western Bluebird abundance increased tenfold. Omnivorous and granivorous bird species (potential pest species) abundance remained constant, suggesting low risk of addition of nest boxes. Plots with nest box addition had 2.4 times more live beet armyworm removal. Further, immediately below nest boxes, removal was 3.5 times higher than in the control (Jedlicka et al., 2011). This study suggests addition of nest boxes for insectivorous birds may be an important part of Integrated Pest Management.
Habitat during the Growing Season
Habitat usage varies throughout the range, but typical habitats are open coniferous and deciduous forests, forest edges, farms, and orchards. In the Willamette Valley, Western Bluebirds are common in open country with scattered trees and orchards, whereas in the eastern Cascades, they are more common in Douglas fir (Pseudotsuga menziesii) and open pine forests (Gilligan et al., 1994). In southern California, Western Bluebirds are primarily found in open oak woodlands and coniferous forests, and rarely in areas with large row crop fields (Garrett and Dunn, 1981).
Management
Results from the North American Breeding Bird Survey indicate a 0.58% range-wide increase from 1966-2012 (Sauer et al., 2014); however, in the Northern Pacific Rainforest, there was a -1.25% decline from 1966-2012 (Sauer et al., 2014). Western Bluebirds are a State Monitor Species in Washington State (Washington Department of Fish and Wildlife; 2017) and are listed as Vulnerable in Oregon (Oregon Department of Fish and Wildlife, 2008). The most important likely contributors are loss of suitable nest sites and foraging areas due to logging, fire suppression, grazing, and urbanization (Herlugson, 1975; Brawn and Balda, 1988). The Western Bluebird is a secondary cavity nester, meaning it requires cavities excavated by other species, and relies on availability of snags, large living trees, or nest boxes (Guinan et al., 2008). Proposed measures include controlled and natural burning, prohibition of snag removal, and preservation of old, partially dead trees (Herlugson, 1975). An estimated 57% of the Washington Western Bluebird population lives on private land (Cassidy and Grue, 2000), suggesting the importance of private landowners providing suitable habitat for this species. Western Bluebirds compete with other native species like the Violet-green Swallow (Tachycineta thalassina) for nest sites, along with invasive House Sparrows (Passer domesticus) and European Starlings (Sturnus vulgaris) (Gillis, 1989). Instructions on deterrence and removal of invasive species can be found here. Bluebird nest-box trails have been implemented to add and monitor nest boxes (Fig. 7). Success is limited by competition with other bird species, but the numbers of nest boxes used by bluebirds has increased markedly since the program began (Guinan et al., 2008). Instructions on nest construction and placement can be found here.

Figure 7. Western Bluebird emerging from a Bluebird Trail nest box. Photo credit: Mick Thompson, Western Bluebird, CC Attribution-NonCommercial 2.0
More Resources
The Cornell Lab of Ornithology (birds.cornell.edu) supports a great citizen scientist network with detailed information on nest construction and placement (nestwatch.org), recommendations on attracting species of interest (content.yardmap.org), and range information (ebird.org). The lab offers many opportunities for the public to get involved with scientific data collection through Project Feederwatch (feederwatch.org), eBird (eBird.org), and Nestwatch (nestwatch.org). Basic species information can be found at allaboutbirds.org, and the Merlin Bird ID app can aid in field identification.
References and Citations
- Bael, S.A.V., S. M. Philpott, R. Greenberg, P. Bichier, N. A. Barber, K. A. Mooney, and D. S. Gruner. 2008. Birds as predators in tropical agroforestry systems. Ecology 89:928—934. Available online at: http://dx.doi.org/10.1890/06-1976.1 (verified 18 April 2017).
- Beal, F.E.L. 1915. Food of the robins and bluebirds of the United States. Bulletin of the United States Department of Agriculture 171.
- Brawn, J. D., and R. P. Balda. 1988. Population biology of cavity nesters in Northern Arizona: Do nest sites limit breeding densities? The Condor 90:61—71. Available online at: http://www.jstor.org/stable/1368434 (verified 10 January 2017).
- Cassidy, K. M., and C. E. Grue. 2000. The role of private and public lands in conservation of at-risk vertebrates in Washington State. Wildlife Society Bulletin 28:1060—1076. Available online at: http://www.jstor.org/stable/3783867 (verified 10 January 2017).
- De Graaf, R. M., N. G. Tilghman, and S. H. Anderson. 1985. Foraging guilds of North American birds. Environmental Management 9:493—536. Available online at: http://dx.doi.org/10.1007/BF01867324 (verified 10 January 2017).
- Duckwork, R. A., and A. V. Badyaev. 2007. Coupling of dispersal and aggression facilitates the rapid expansion of a passerine bird. Proceedings of the National Academy of Sciences of the United States of America 104:15017—15022. Available online at: http://www.pnas.org/content/104/38/15017 (verified 10 January 2017).
- Garrett, K., and J. Dunn. 1981. Birds of southern California: Status and distribution. Los Angeles Audubon Society, Los Angeles, CA.
- Gilligan, J., D. Rogers, M. Smith, and A. Contreras. 1994. Birds of Oregon: Status and distribution. Cinclus Publications, McMinnville, OR.
- Gillis, E. 1989. Western Bluebirds, Tree Swallows, and Violet-green Swallows west of the Cascade Mountains in Oregon, Washington, and Vancouver Island, British Columbia. Sialia 11:127—130.
- Guinan, J. A., P. A. Gowaty, and E. K. Eltzroth. 2008. Western Bluebird (Sialia Mexicana). In P. Rodewald (ed.) The Birds of North America. Cornell Lab of Ornithology, Ithaca, NY. Available online at: https://birdsna.org/Species-Account/bna/species/wesblu (verified 10 January 2017).
- Herlugson, C. J. 1975. Status and distribution of the Western Bluebird and the Mountain Bluebird in the state of Washington. Master's Thesis, Washington State University, Pullman, WA.
- Herlugson, C. J. 1982. Food of adult and nestling Western and Mountain Bluebirds. The Murrelet 63:59—65. Available online at: http://www.jstor.org/stable/3533829 (verified 10 January 2017).
- Jedlicka, J. A., R. Greenberg, and D. K. Letourneau. 2011. Avian conservation practices strengthen ecosystem services in California vineyards. PLoS ONE 6: e27347. Available online at: http://dx.doi.org/10.1371/journal.pone.0027347 (verified 10 January 2017).
- Jedlicka, J. A., A. E. Vo, and R.P.P. Almeida. 2017. Molecular scatology and high-throughput sequencing reveal predominately herbivorous insects in the diets of adult and nestling Western Bluebirds (Sialia mexicana) in California vineyards. The Auk 134:116—127. Available online at: http://dx.doi.org/10.1642/AUK-16-103.1 (verified 16 January 2017).
- Mols, C. M., and M. E. Visser. 2002. Great Tits can reduce caterpillar damage in apple orchards. Journal of Applied Ecology 39:888—899. Available online at: http://onlinelibrary.wiley.com/doi/10.1046/j.1365-2664.2002.00761.x/full (verified 13 April 2017).
- Oregon Department of Fish and Wildlife [Online]. Wildlife Division/Conservation/Species/Sensitive species. Available at: http://www.dfw.state.or.us/wildlife/diversity/species/sensitive_species.asp (verified 19 April 2017).
- Sauer, J. R., J. E. Hines, J. E. Fallon, K. L. Pardieck, D. J. Ziolkowski, Jr., and W. A. Link. 2014. The North American breeding bird survey, results and analysis 1966—2013. Version 01.30.2015 USGS Patuxent Wildlife Research Center, Laurel, MD. Available online at: https://www.mbr-pwrc.usgs.gov/bbs/ (verified 2 January 2017).
- Washington Department of Fish & Wildlife [Online]. Washington State species of concern lists. Available at: http://wdfw.wa.gov/conservation/endangered/status/SM/ (verified 10 January 2017).



